|
The older IUPAC system used Roman numerals together with letters to distinguish between the left (A) and right (B) side of the periodic table.Three systems have been used to number families and groups: Recognizing Families on the Periodic TableĬolumns of the periodic table typically mark groups or families. Noble Gases: - Group 18 (VIIIA) - 8 valence electrons.Halogens: - Group 17 (VIIA) - 7 valence electrons.Oxygen Group or Chalcogens: - Group 16 (VIA) - 6 valence electrons.Nitrogen Group or Pnictogens: - Group 15 (VA) - 5 valence electrons.Carbon Group or Tetrels: - Group 14 (IVA) - 4 valence electrons.Boron Group or Earth Metals: Group 13 (IIIA) - 3 valence electrons.Transition Metals: Groups 3-12 - d and f block metals have 2 valence electrons.Alkaline Earth Metals: Group 2 (IIA) - 2 valence electrons.Alkali Metals: Group 1 (IA) - 1 valence electron.Many chemists and chemistry textbooks recognize five main families:Īnother common method of categorization recognizes nine element families: However, there are different ways of categorizing elements into families. Because element properties are largely determined by the behavior of valence electrons, families and groups may be the same. :max_bytes(150000):strip_icc()/PeriodicTableCharge-BBG-58b5c80a3df78cdcd8bbb6c8.png)
Element groups, on the other hand, are collections of elements categorized according to similar properties. The characteristics of the elements in these families are determined primarily by the number of electrons in the outer energy shell. 
Elements are classified into families because the three main categories of elements (metals, nonmetals, and semimetals) are very broad. This behavior is in sharp contrast to that of the p-block elements, where the occurrence of two oxidation states separated by two electrons is common, which makes virtually all compounds of the p-block elements diamagnetic.ĭue to a small increase in successive ionization energies, most of the transition metals have multiple oxidation states separated by a single electron.Element families are indicated by numbers located at the top of the periodic table.Īn element family is a set of elements sharing common properties. The occurrence of multiple oxidation states separated by a single electron causes many, if not most, compounds of the transition metals to be paramagnetic, with one to five unpaired electrons. Because of the slow but steady increase in ionization potentials across a row, high oxidation states become progressively less stable for the elements on the right side of the d block. Manganese, for example, forms compounds in every oxidation state between −3 and +7. The relatively small increase in successive ionization energies causes most of the transition metals to exhibit multiple oxidation states separated by a single electron. Thus all the first-row transition metals except Sc form stable compounds that contain the 2+ ion, and, due to the small difference between the second and third ionization energies for these elements, all except Zn also form stable compounds that contain the 3+ ion. This in turn results in extensive horizontal similarities in chemistry, which are most noticeable for the first-row transition metals and for the lanthanides and actinides. The similarity in ionization energies and the relatively small increase in successive ionization energies lead to the formation of metal ions with the same charge for many of the transition metals. 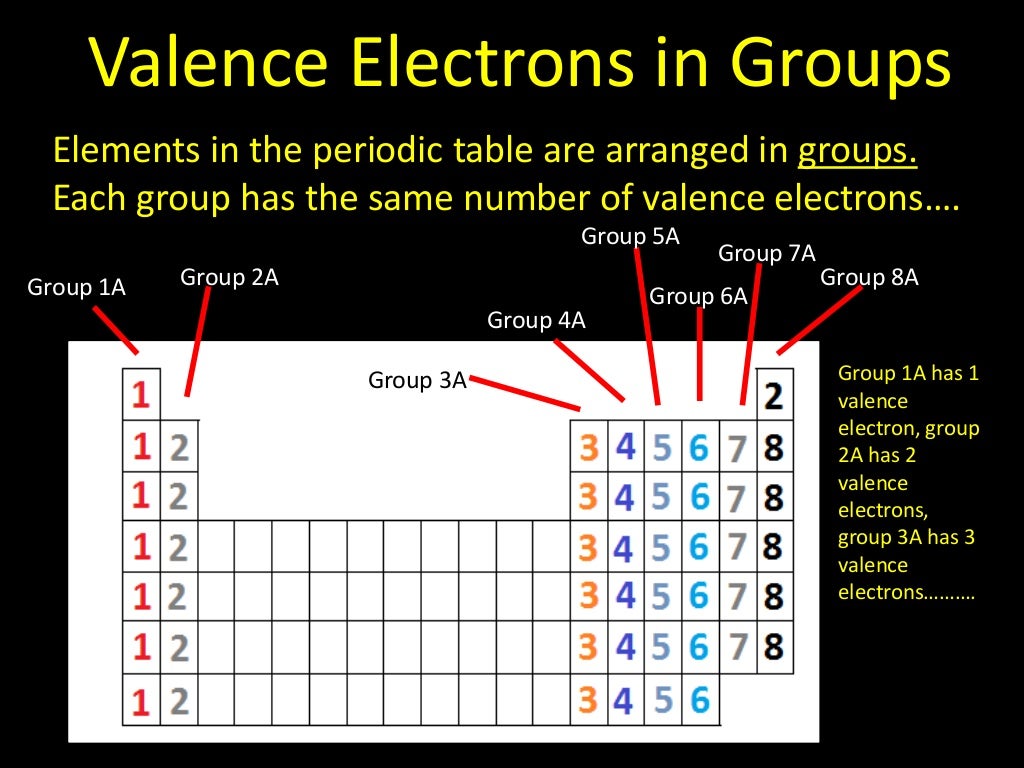 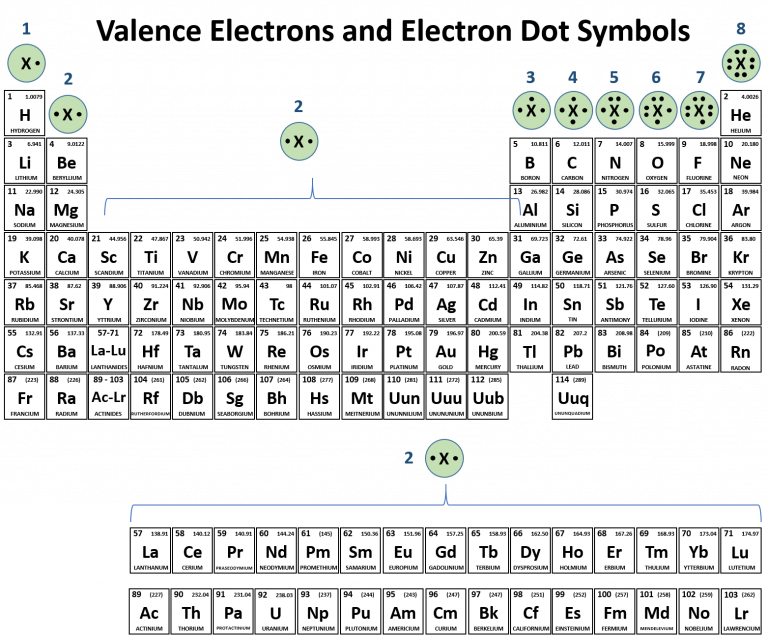
Trends in Transition Metal Oxidation States As a result, the metals in the lower right corner of the d block are so unreactive that they are often called the “noble metals.” The electronegativity of the elements increases, and the hydration energies of the metal cations decrease in magnitude from left to right and from top to bottom of the d block. \): Some Trends in Properties of the Transition Metals.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
