In addition, the alcohol equilibration step allows the gel to adjust to its final size before transfer. Equilibrating the gel in alcohol removes contaminating electrophoresis buffer salts and prevents an increase in the conductivity of the transfer, which can increase the amount of heat generated. If using a gel chemistry other than Tris-acetate, adding a quick alcohol equilibration step before transfer can greatly enhance the transfer of HMW proteins when not using the ideal gel chemistry. 
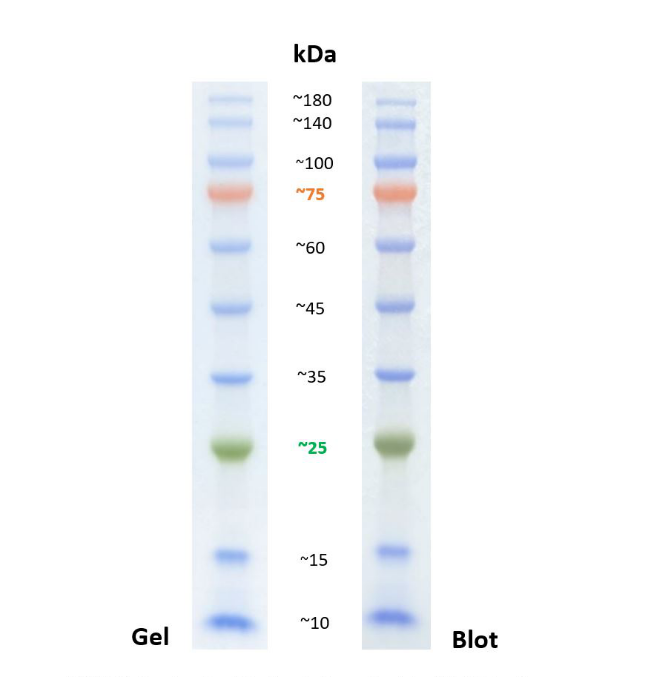
A32735) at a dilution of 1:5,000 for one hour at room temperature. After overnight incubation, the membranes were washed in TBST and probed with Goat anti-Rabbit (H+L) Highly Cross-Adsorbed Secondary Antibody, conjugated to Alexa Fluor Plus 800 (Cat. 37565) at room temperature and then probed overnight at 4☌ with an EGFR polyclonal antibody (Cat. Membranes were blocked for 30 minutes with Blocker FL Fluorescent Blocking Buffer (Cat. Proteins were transferred with the iBlot 2 Gel Transfer Device onto nitrocellulose membranes (Cat. EA03752BOX) or onto a Novex 4–20% Tris-glycine gel, WedgeWell format (Cat. Method: Western blot analysis of EGFR was performed by loading serially diluted A431 cell lysate onto a NuPAGE 3–8% Tris-acetate gel (Cat. Western blotting analysis of EGFR expression in A431 lysates transferred from an Novex 4–20% Tris-glycine gel and a NuPAGE 3–8% Tris-acetate gel using the iBlot 2 Gel Transfer Device. As seen in Figure 2, better transfer is seen using a Tris-acetate gel over a 4–20% Tris-glycine gel-9 ng visualized when a Tris-acetate gel was used vs 750 ng visualized when a Tris-glycine gradient gel was used in targeting ~190 kDA protein epidermal growth factor (EGFR).įigure 2. A comparison of HMW protein separation using different gel chemistries and gradients shows the best separation and resolution of HMW proteins can be accomplished with a 3–8% Tris-acetate gel ( Figure 1B). The open matrix structure that allows the HMW proteins to migrate farther through the gel allows better transfer of the HMW proteins out of the gel leading to increased transfer efficiencies and higher sensitivity. By using a Tris-acetate or low gradient Bis-Tris or Tris-glycine gel, HMW proteins can migrate further through the gel, allowing increased distance between protein bands. Proteins >200 kDa are compacted into a very narrow region at the top of the running portion of the gel, leading to poor resolution of protein bands ( Figure 1). While 4–20% Tris-glycine gradient gels are very popular because of their ability to separate a broad range of proteins (20–200 kDa), they are not recommended for separation of HMW proteins. 
When targeting HMW proteins for your transfer, it is best to use a Tris-acetate gel or low percentage Bis-Tris or Tris-glycine gel. 
The ladder's probably fine, but if these are cell lines or mouse tissue you should bump it up a bit (assuming your 1Abs are high affinity and specificity for your target and are generally doing their job well).Choosing the right gel is a key factor in the successful transfer of HMW proteins. Also, are you loading enough protein per well? The signal looks extremely faint and I can really only make out 2 sample lanes. Then, I'd work backwards and make sure it's not my ECL reagent (is it fresh?), my 2Abs (is the concentration high enough to get a good signal?), my 1Abs (are these high affinity enough for my target? What happens if I double or half my usual concentration?), my transfer (did I transfer for long enough and did all my protein get transfered? I'd do a Ponceau or check that all dye left the gel in the case of visual dyes in the molecular ladder), and finally my gel running settings. Because it's easy, I would maybe first play around with the length of exposure on the chemidoc and just make sure the issue isn't there first. Voltage during the run, if too high, can definitely lead to smearing in the high molecular weight range, band "smiling" or other artifacts. You may want to see if lowering the voltage a bit or switching running buffers makes a difference for the stacking and tightness of the ladder bands. Okay that helps a bit! Regarding the ladder, I assume it's detected via HRP? Or is it a proprietary BIO-RAD technology that allows it to be detected in some other wavelength range? IMO it's a little blurry and it could be stronger.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
